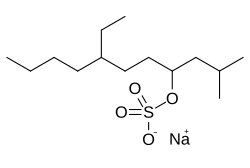
Overview
Molecular formula: C₁₄H₂₉NaO₄S
Synonyms: STDS, Sodium Tetradecyl Sulfate, STS
Molecular weight: 316.43 g/mol
Appearance: White to off-white solid or defined pharmaceutical formulation
Solubility: Soluble in water
CAS-Number: 139-88-8
Sodium Tetradecyl Sulfate (STDS) is a well-established active pharmaceutical ingredient used in specialized medical applications. Its use requires consistent quality, controlled specifications, and reliable analytical characterization. ChemCon stands for GMP-compliant manufacturing of high-quality specialty substances and supports customers with robust documentation and regulatory expertise.
Medical Applications
Sodium Tetradecyl Sulfate (STDS) is used as a sclerosing agent and is primarily applied in the treatment of varicose veins as part of sclerotherapy. In addition, STDS is used in further vascular applications where targeted sclerosis of vessels is therapeutically indicated.
Regulatory Status:
FDA approvals (USA) as well as national approvals in Europe for applications in sclerotherapy, particularly for the treatment of varicose veins.
Mechanism of Action
- STDS acts as a sclerosing agent that induces a targeted reaction at the vascular endothelium following administration. This results in the occlusion of the treated vessel and supports the treatment of varicose veins within established medical procedures.
Advantages of STS in Therapy
- Proven mechanism of action as a sclerosing agent
- Targeted local effect on the vascular endothelium
- Extensive clinical experience in sclerotherapy
- Used in approved medicinal products across various markets
- Suitable for different formulations depending on the application
ChemCon STDS
At ChemCon, we stand for the highest quality. That's why we offer STS in up to injection grade. In our laboratories, certified by the regulatory authorities and the FDA, we meet the strictest quality requirements in the production of STS.
ChemCon quality
At ChemCon, we stand for the highest quality in the production of our products, including STS. Our production processes meet the strictest international standards to ensure that all products, including Sodium Tetradecyl Sulfate in injection grade, meet the highest requirements. We provide complete documentation and comprehensive regulatory support to help our customers comply with all relevant regulations. Through continuous quality controls and the certification of our laboratories by the regulatory authorities and the FDA, we ensure that every product meets the highest quality standards.
Downloads
Here you can find more information
![ChemCon Company Brochure Product Examples [Translate to Englisch:] ChemCon products](/fileadmin/_processed_/a/5/csm_ChemCon_Products_b2812b25f2.png)
ChemCon high quality APIs
![[Translate to Englisch:] ChemCon company brochure](/fileadmin/_processed_/2/2/csm_ChemCon_Company_Brochure-2024_web_144dpi_75__53bcee528e.jpg)
ChemCon company brochure
Sources:
U.S. Food and Drug Administration (FDA): Sotradecol® (Sodium Tetradecyl Sulfate Injection), https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=f1756c28-dcd2-4b49-be62-07ca20682018, Mar 2026
Electronic Medicines Compendium (emc): Fibrovein® Sodium Tetradecyl Sulfate, https://www.medicines.org.uk/emc/product/1199/smpc, Mar 2026
Wikipedia: Sodium tetradecyl sulfate, https://en.wikipedia.org/wiki/Sodium_tetradecyl_sulfate, Mar 2026 / CC-BY-SA 4.0